Microneedle biosensor could provide real-time measurements of antibiotic concentrations
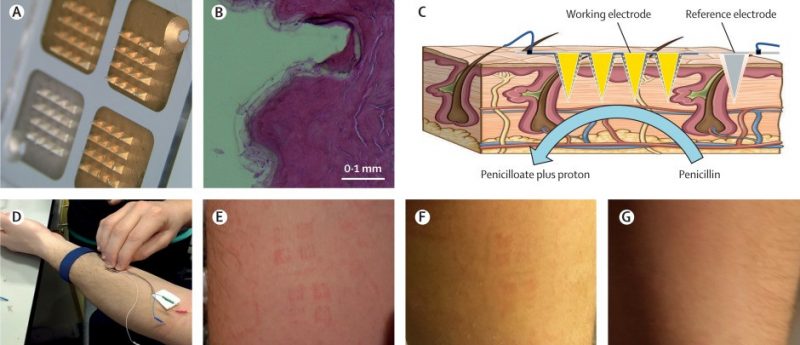
Researchers based at Imperial College London (UK) have released the findings of a first-in-human, proof-of-concept trial published in The Lancet Digital Health. The trial reports that a microneedle biosensor applied to the forearm was able to measure extracellular fluid penicillin concentrations demonstrating results comparable to microdialysis and serum concentration measurement in blood samples.
As the understanding of antimicrobial PK/PD profiles improves and the multitude of pathophysiological changes and individual differences becomes more apparent, researchers are becoming increasingly interested with the prospect of customized drug dosing.
The findings of the study could represent a step towards customized drug dosing, where real-time penicillin drug concentrations can be measured using a small and easy to wear microneedle biosensor and drug dosing can be changed accordingly.
In the study, a microneedle biosensor (1.5 cm2) evaluated the extracellular fluid of ten healthy volunteers in real-time for up to 6 hours while the patient was dosed at steady state with oral phenoxymethyl penicillin. Drug concentrations were also measured by standard techniques at the same time points for comparison.
The data demonstrated that the microsensors showed similar free drug area under the curve when compared with serum area under the curve measured by current standard techniques. The PK profiles of the microsensors were similar to the current standard of microdialysis methods of extracellular fluid monitoring.
The early study has a number of limitations, including small sample size and the use of one drug on healthy patients. Further investigation is required to show whether this technology has the potential to change antimicrobial treatment or become the standard technique in the clinic.
Co-author Anthony Cass (Department of Chemistry, Imperial College) explained: “This small, early-stage trial has shown that the sensor technology is as effective as gold standard clinical analysis in detecting changes to the concentrations of penicillin in the human body. When further developed, this technology could prove critical for the monitoring and treatment of patients with severe infections. More widely it could be used to monitor many other drugs and personalize treatment in many diseases.”
Sources: Rawson TM, Gowers SAN, Freeman DME et al. Microneedle biosensors for real-time, minimally invasive drug monitoring of phenoxymethylpenicillin: a first-in-human evaluation in healthy volunteers. Lancet Dig. Health. doi:10.1016/S2589-7500(19)30131-1 (2019)(Epub ahead of print); www.imperial.ac.uk/news/193146/microneedle-biosensors-accurately-detect-patient-antibiotic/
