Poster: novel approach to immunogenicity testing for therapeutics with high prevalence of pre-existing antibodies
Abstract:
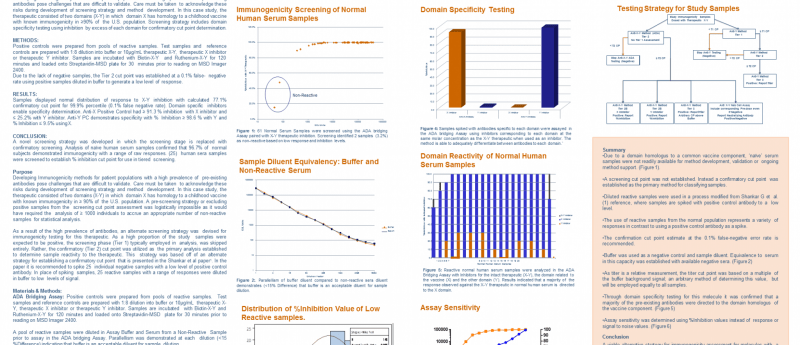
Developing immunogenicity methods for patient populations with a high prevalence of pre-existing antibodies pose challenges that are difficult to validate. Care must be taken to acknowledge these risks during development of screening strategy and method development. In this case study, the therapeutic consisted of two domains (X-Y) in which domain X has homology to a childhood vaccine with known immunogenicity in ≥90% of the US. population. Screening strategy includes domain specificity testing using inhibition by excess of each domain for confirmatory cut point determination.
To download this poster, please click here!
For more information on Eurofins, please click here!