Automation and Logistics

J oseph Tweed is a bioanalytical scientist working at Pfizer Inc. located in Groton, CT, USA. Joseph has over 18 years of multi-disciplinary experience in the areas of drug discovery, drug development and pharmacology research. He is responsible for developing and directing automated bioanalytical techniques that can improve the quality and the efficiency of day-to-day laboratory operations. He is actively engaged in pursuing automated innovations for use in the regulated bioanalysis of small-molecules, antibody-drug conjugates (ADCs) and biomarkers. Joseph received his B.S. from Drexel University, a M.S. from Thomas Jefferson University and his M.B.A from the University of New Haven.
oseph Tweed is a bioanalytical scientist working at Pfizer Inc. located in Groton, CT, USA. Joseph has over 18 years of multi-disciplinary experience in the areas of drug discovery, drug development and pharmacology research. He is responsible for developing and directing automated bioanalytical techniques that can improve the quality and the efficiency of day-to-day laboratory operations. He is actively engaged in pursuing automated innovations for use in the regulated bioanalysis of small-molecules, antibody-drug conjugates (ADCs) and biomarkers. Joseph received his B.S. from Drexel University, a M.S. from Thomas Jefferson University and his M.B.A from the University of New Haven.
At its core, the bioanalytical discipline strives to deliver timely quantitative data to drug discovery and development project teams. Within the discipline there are many considerations that affect how bioanalytical data is generated and subsequently applied for informative decision making related to drug portfolio progression. Over the course of the past 20 years many scientific and technology advances in drug research and development (R&D) have resulted in new and interesting bioanalytical challenges. Fortunately, these advances have positively impacted the evolution of the bioanalytical discipline which, in turn, has resulted in improved assay quality, reliability and throughput.
One area that has helped advance the bioanalytical discipline through the evolving R&D landscape has been the continued use of automation in bioanalysis. The value of automation has been well recognized within the literature and continues to be an area of continued expansion and innovation. As a result, automation continues to be broadly and successfully applied within the bioanalytical discipline. Some routine examples include automated biospecimen sample organization and storage systems, fully integrated robotic liquid handling sample preparation platforms and automated data collection, processing and report generation.
Fully leveraging automation in the bioanalytical laboratory requires considerations that should include biospecimen sample logistics and lifecycle management (collection, receipt, storage and organization, analysis, disposal), as well as the strategic management of the corresponding bioinformatic data (subject/animal, position, barcodes, etc.) and traveling with the biospecimen sample. A few examples are provided below to detail how these considerations can be complementary to automation, as well as some new approaches that will enable more streamlined and integrated approaches to bioanalytical data generation.
Biospecimen sample collection:
Significant changes have been observed in biospecimen sample collection over the past two decades. For example, significant resources have been applied to some new and interesting sample collection procedures such as DBS and related serial sampling approaches such as capillary microsampling (CMS) [1] and volumetric absorptive microsampling (VAMS) [2,3]. One particular emerging approach developed by Fluisense, Inc. uses a small, wearable microfluidic sampling device (fluispotter®) to serially collect and store DBS samples over a 24 hour period from small rodents or other animal species used in drug discovery or safety studies. Unique to the fluispotter® and the DBS samples it generates is its ability to collect a blood sample simultaneously with a given internal standard at the time of blood sampling and subsequent application to the solid fiber substrate (Figure 1). As serial samples are collected and stored within the micro-device, barcode information can be associated with each DBS sample collected, providing reliable tractability and tracking throughout the collection and subsequent analysis of the DBS samples.
Logistics and biospecimen sample management:
Arguably, the impact of change within the bioanalytical discipline may not be as drastic in the routine logistics and management of biospecimen samples. By and large we still see the same processes and procedures used in the shipment and accounting of biospecimen samples that were in place over this same time frame mentioned above. Fortunately, there is a clear understanding in how sound biospecimen sample management and logistics processes/procedures ensure proper biospecimen chain-of-custody, compliance and sample integrity. For instance, the bioanalytical discipline has seen an almost universal adaptation of laboratory information management systems (LIMS) and an increased use in electronic laboratory notebook (ELNs) for discovery and development data collection. However, the topic of sample management and logistics often does not receive the necessary attention required and in some instances may seem antiquated in relation to current tracking and logistics technologies as they exist today.
The physical analysis of biospecimen samples for any given study is impacted by the quality of the logistical procedures and sample management processes both downstream and upstream of the actual data collection. Organizations that carefully consider biospecimen management and logistics into their preclinical and clinical workflows will positively impact the timeliness and quality of the bioanalytical data generated. Some organizations may take logistical considerations related to biospecimen sample management for granted, and therefore do not identify logistics as an area for innovative approaches to optimize biospecimen lifecycle management. However, organizations that appropriately leverage basic technology like 1 and 2-dimensional barcoding through a biospecimen sample’s lifecycle, facilitate overall quality and efficiency through improved tractability, compliance and integration into automation hardware and software [4,5].
Pairing logistical considerations to routine laboratory tasks should also be considered. Obviously, as technology evolves so does the opportunity to expand the utility and value in how automation can potentially be applied and leveraged within the laboratory to achieve the anticipated return on investment. One area in which automation has perhaps has had a less of an impact is in the routine preparation of solutions and solvents used in large and small molecule bioanalytical assays (i.e. buffers, mobile phases, solid phase extraction solvents, etc.). With the introduction of a new automated platform called the Revo from LabMinds, Inc. (Figure 2) there is now an option to prepare protocol-driven solutions remotely with full tractability and consistent quality.
The Revo platform is able to customize solution and solvent preparation by combining solid chemical addition, mixing, pH adjustments, vacuum filtering and GLP compliant labeling into one automated production system. It can certainly be argued that this type of automated platform is a luxury, as these repetitive and mundane tasks are essentially a resource (time) expense to any organization. However, when we standardize approaches such as these and add full traceability, consistency and programmable scheduling the value of automating these types of tasks becomes more tangible. Platforms such as these remain untested within the bioanalytical discipline. Nonetheless, the Revo and other competitors that may emerge offer a new and potentially more reliable, compliant and convenient automated solution to consider.
In closing, the bioanalytical discipline continues to use automation in new and interesting ways. Thinking about bioanalytical approaches to automation should not only include assay method development and design, but also the supporting logistics and biospecimen management practices routinely used in the laboratory. As a discipline, we should continue to build upon the success within the automation space and strive to integrate logistical and biospecimen lifecycle management considerations into our workflows for sustained success in the future.
References
- Korfmacher W, Fitzgerald M, Luo Y, et al.Capillary microsampling of whole blood for mouse PK studies: an easy route to serial blood sampling. Bioanalysis 7(4), 449–461 (2015).
- Denniff P, Spooner N. Volumetric absorptive microsampling: a dried sample collection technique for quantitative bioanalysis. Anal. Chem. 86(16), 8489–8495 (2014).
- Grafmuller L, Tweed JS, Gu Z, Wallace M, Ramanathan R, Steenwyk R. The evaluation and development of automated workflows in blood, plasma and urine using volumetric absorptive microsampling (VAMS). Presented at: 63rd Annual ASMS Conference. MO, USA, 31 May – 4 June 2015.
- Tweed JA, Walton J, Gu Z. Automated supported liquid extraction using 2D barcode processing for routine toxicokinetic portfolio support. Bioanalysis 4(3), 249–262 (2012).
- Tweed JA. Automation in LC‐MS Bioanalysis. In: Handbook of LC-MS Bioanalysis: Best Practices, Experimental Protocols, and Regulations. Li W, Zhang J, Tse FLS (Eds). Wiley, NJ, USA, 275–295 (2013).
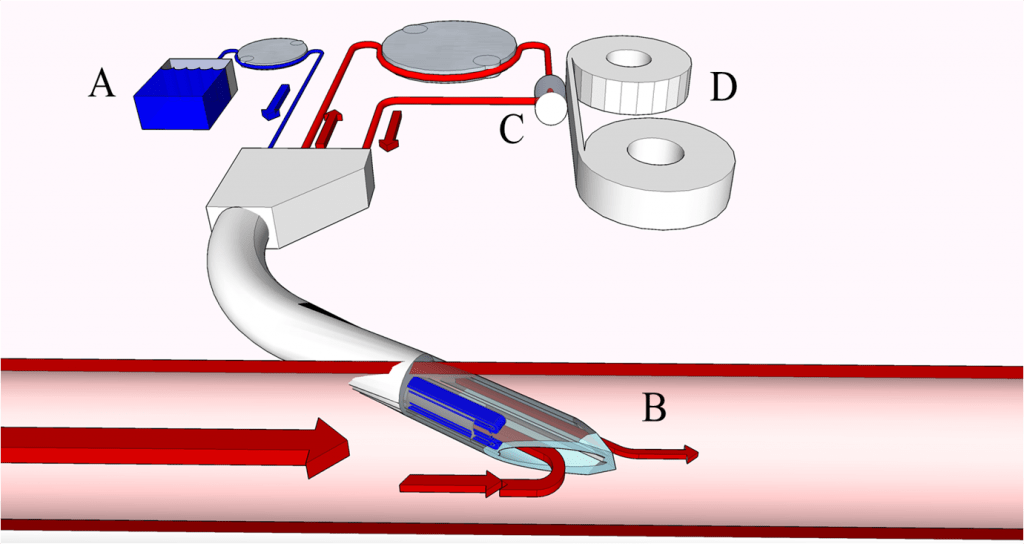
Figure 1: Schematic representation of the internal operations of the fluispotter®, a fully portable and automated blood sampling system designed and manufactured by Fluisense®. At time of sampling, an internal standard solution from reservoir (A) is mixed with venous blood sampled from the cannula (B). The fortified blood sample is measured via a micro-peristaltic pump and quantitatively spotted (C) onto a blood spot reel (D) for sequential blood sampling. Image adapted from Fluisense ApS, Denmark.

Figure 2: Photo of the Revo automated liquid solution production system developed and manufactured by LabMinds, Ltd. The Revo platform centralizes and automates a variety of procedural laboratory task such as solution, buffer, mobile phase preparation with fully integrated data collection and traceability into electronic laboratory notebooks. Copyright and Revo image adapted from LabMinds, Ltd, Boston, MA with permission.