Hybrid LBA/LC–MS survey infographic

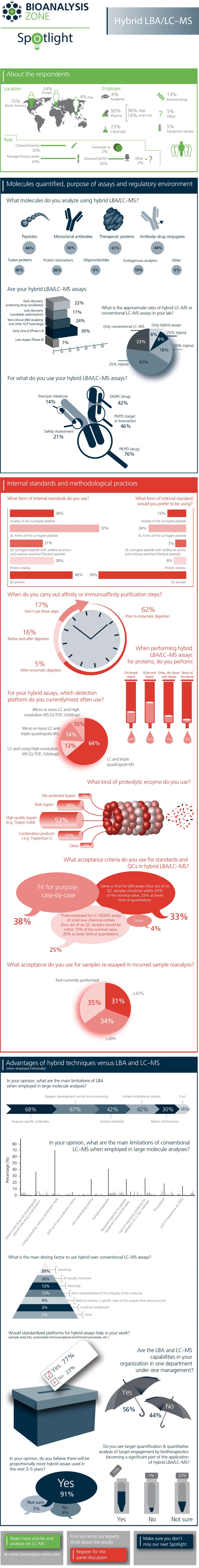
As part of our Spotlight on Hybrid LBA/LC–MS, we carried out a survey to gain insight into the molecules you analyze using this technology and the regulatory environment you work in. The survey addressed methodological practices followed in LBA/LC–MS, including acceptance criteria used for drug analysis and much more.
We now have the results of our survey! We had a great response, so thank you to everyone who took part. Check out our informative infographic below.
Do not forget to attend our online panel discussion, taking place on Thursday June 23rd, with Rand Jenkins (PPD® Laboratories), Carsten Krantz (Novartis), Suma Ramagiri (SCIEX), Jian Wang (Bristol-Myers Squibb) and Megan Wiberg (PPD® Laboratories) providing expert analysis of the survey results.