11th WRIB poster award winner: John Lowe

The Bioanalysis Zone and Bioanalysis team attended this year’s 11th Workshop on Recent Issues in Bioanalysis (WRIB; LA, USA), 3–7 April 2017. We were particularly proud to support and present the WRIB Poster Awards to Annelies Codden, John Lowe and Neal Simmons. Here, we feature the winning poster from John Lowe (Genentech) as well as an exclusive interview.
Click the image below, or click here to view the winning poster:
What the judges had to say
“The authors provided a very nice description of the technology and the practical applications. This poster will help inform the bioanalytical community on the feasibility of the Simple Plex multiplexing system in a challenging rare matrices setting.” – Heather Myler (Principal Scientist, Analytical and Bioanalytical Development, Britsol-Myers Squibb)
Interviw with John Lowe
I am currently a Principal Scientific Researcher working in the BioAnalytical Sciences Department at Genentech Inc. (CA, USA). I received my BA in biological sciences from University of California, Berkeley and MS in Clinical Sciences from San Francisco State University. I joined Genentech Inc. in 1990 after working for 4 years at University of California, San Francisco where I completed my Master thesis. I specialize in developing PK, ADA, and biomarker assays to support pre-clinical and clinical studies for large molecules that are in late research and development stages. My research interests include evaluation of new technologies and application of novel bioanalytical methods in assay development to support pre-clinical and clinical studies.
1Please can you explain about your role at Genentech?
I am currently a Principal Scientific Researcher working in the BioAnalytical Sciences Department at Genentech Inc.. My main responsibility is to develop rugged analytical methods that include PK, ADA, and biomarker assays to support pre-clinical and clinical studies for large molecules that are in late research and development stages.
2How does it feel to win the 11th WRIB Poster Award?
I felt extremely privileged and thought it was a tremendous honor for our poster to be recognized in this prestigious meeting. I would like to thank all of my coauthors and colleagues who had collaborated and offered support for this project.
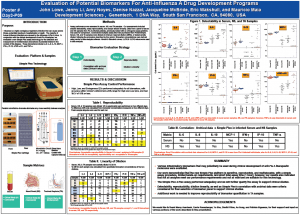
3 Tell us a little about the work on which the Poster was based?
In conjunction with Genentech’s clinical development of anti-Flu A therapies, we evaluated Simple Plex assay technology for the detection and measurement of 8 inflammatory biomarkers of potential interest in the context of this disease. Assay performance was assessed in three matrices of pharmacological interest including serum, nasal swab (NS), and tracheal aspirate (TA) samples. Our assessments included analyte detectability, assay reproducibility, and sample dilution linearity. Sample analysis was performed in the Simple Plex 4-plex assay for a panel of 8 biomarkers: IL-6, IL-8, IL-10, IP-10, MCP-1, IFN-γ, TNF-α, and TNF-α-R1. Results for controls of all biomarkers were accurate and reproducible. The assay also showed acceptable detectability, reproducibility, and linearity of dilution for serum and NS samples. In addition, Simple Plex 4-plex assay results showed good correlation with archival data for IL-8, MCP-1, and IP-10, when measured in serum as well as in NS samples.
4 What were the key conclusions from your research?
Various inflammatory biomarkers that may potentially be used during clinical development of anti-Flu A therapeutic molecules were evaluated. Our work demonstrated that the Simple Plex platform is sensitive, reproducible, and multiplexable, with a simple assay procedure, limited hands-on requirements, and short total assay time (1 hour); however, our results also indicated that not all analytes meet our performance requirements and not all matrices are suitable for this technology. The Simple Plex 4-Plex assay performed adequately and we will further qualify the assay to support clinical studies.
5What are you looking forward to working on over the next year?
We showed that with TA samples, detectability and dilution linearity were acceptable, whereas reproducibility was not. This lack of reproducibility with TA samples was likely due to the heterogeneous nature of the samples, which may be mitigated by implementing sample-pretreatment methods. Therefore, additional work is needed to improve reproducibility of biomarker quantitation in TA samples.
You can view posters and interviews from the other award winners here.