COVID-19 testing: potential challenges and solutions

As the COVID-19 pandemic extends its reach worldwide, the demand for available, accurate testing will grow exponentially. Science News reports that COVID-19 testing has been available in limited quantities from the Centers for Disease Control (CDC), but production has not been able to keep up with demand. The CDC plans to make millions of tests available as soon as possible; in the meantime, private labs have ramped up their capabilities to create and analyze tests as well as to develop potential vaccines.
Testing process
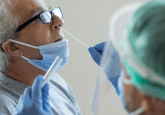
Administered by healthcare professionals — some in a drive-up capacity, in order to limit person-to-person exposure — tests for COVID-19 use respiratory samples obtained via nasopharyngeal (NP) or oropharyngeal (OP) aspirates or washes, NP or OP swabs, or bronchoalveolar lavage (BAL).
Scientists then purify the virus’s genetic material from the sample, a time- and labor-intensive step that has the potential to create testing bottlenecks. All tests for COVID-19 use a technique called a polymerase chain reaction (PCR), which can detect tiny amounts of viral genetic material. SARS-CoV-2, the virus that causes COVID-19, has single-strand RNA as its genetic material. This RNA is must be copied into double-strand DNA in order to be tested via PCR, a process that can take up to 30 minutes. The CDC’s test scans for two of the virus’s genes, and the World Health Organization’s (WHO) test scans for three of the genes. The entire testing protocol can take up to 3 hours, which poses challenges in terms of volume as demand grows.
Private labs
Most private labs are pursuing testing validation under the FDA’s Emergency Use Authorization process (EUA). This means that new tests can be used with patients as soon as they are validated to the FDA’s standards, after which the lab must file for EUA within 15 days. Additionally, the FDA will allow private companies to market tests to the public without prior approval, according to the Wall Street Journal.
There are numerous challenges for private labs as they work to develop testing capabilities — including staffing shortages, instrument access and supply shortages — as reported in Modern Health. For clinical workflow validation, labs need access to reagents, extraction kits and instruments, PCR systems and control materials. Currently, the supply of materials is not meeting demand — in particular, there is a shortage of attenuated virus and viral genome. Additionally, real-time PCR instrumentation and automated RNA extraction equipment are on backorder from manufacturers.
Vaccine development
According to the World Health Organization, there are currently more than 20 vaccines in development around the globe for the treatment of COVID-19 and multiple therapeutics in clinical trials.
QPS is equipped with an access-restricted bioanalysis lab prepared to analyze COVID-19 clinical trial samples at their campus in Newark, DE, USA. This dedicated lab provides space for drug companies to quickly process samples and deliver results to evaluate the safety and effectiveness of potential vaccines and drug therapies. QPS has decades of experience helping drug companies slow the progress of viral outbreaks and is committed to supporting the life sciences community in fighting COVID-19.
 Since 1995, QPS has provided discovery, preclinical and clinical drug development services. An award-winning leader focused on bioanalytics and clinical trials, QPS is known for proven quality standards, technical expertise, a flexible approach to research, client satisfaction and turnkey laboratories and facilities. For more information, visit www.qps.com or email [email protected].
Since 1995, QPS has provided discovery, preclinical and clinical drug development services. An award-winning leader focused on bioanalytics and clinical trials, QPS is known for proven quality standards, technical expertise, a flexible approach to research, client satisfaction and turnkey laboratories and facilities. For more information, visit www.qps.com or email [email protected].
The opinions expressed in this feature are those of QPS and do not necessarily reflect the views of Bioanalysis Zone or Future Science Group.
Our expert opinion collection provides you with in-depth articles written by authors from across the field of bioanalysis. Our expert opinions are perfect for those wanting a comprehensive, written review of a topic or looking for perspective pieces from our regular contributors.
See an article that catches your eye? Read any of our Expert Opinions for free.