What is mass spectrometry and how does it work?
Mass spectrometry is an analytical technique used to determine molecular mass. It is the analysis of proteins and other molecules involving ionization and measurement of the mass-to-charge (m/z) ratio of the resulting ions, enabling the identification of molecules in pure samples and complex mixtures.
Applications of mass spectrometry
- Pharmaceutical research: identifying active pharmaceutical ingredients, metabolites and impurities.
- Proteomics: studying protein structures, post-translational modifications and protein-protein interactions.
- Environmental sciences: detecting pollutants, toxins and contaminants in air, water or soil.
- Forensic sciences: analyzing drugs or biological samples for criminal investigations.
- Clinical diagnostics: identifying and quantifying molecules in routine clinical tests, disease screening and therapeutic drug monitoring.
Mass spectrometry: step by step
Step 1: Ionization
Mass spectrometry begins with the vaporization of the sample, converting it into a gaseous state. The vaporized sample is introduced into an ionization chamber, where it is bombarded by a stream of electrons, which are emitted from an electrically-heated metal coil. As electrons collide with the sample particles, one or more electrons are “knocked off” the sample particles, resulting in the formation of positively charged ions.
Step 2: Acceleration
The positively charged ions are repelled by the positively charged ionization chamber and are accelerated towards a series of negatively charged slits. This acceleration separates the ions based on their m/z, as lighter ions move faster than heavier ions.
Step 3: Deflection
The stream of positively charged ions enters a magnetic field, where they are deflected based on their m/z. The degree of deflection depends on both the mass and charge of the ion. For example:
- Lighter ions are deflected more as they have less inertia and are more easily influenced by the magnetic field.
- Ions with a higher charge (> +1) experience stronger interactions with the magnetic field, resulting in greater deflection compared to ions with a single positive charge (+1).
- Heavier ions or ions with a lower charge are deflected less due to their greater inertia or weaker interaction with the magnetic field.
Step 4: Detection
The ions pass through the mass analyzer and are detected on the basis of their m/z. When an ion hits the detector, its charge is neutralized as an electron jumps from the detector’s metal surface onto the ion. This generates an electrical current proportional to the ion’s abundance. The resulting data is used to produce a mass spectrum, which displays the m/z values of the detected ions and their relative abundance. This spectrum provides critical information about the composition and structure of the sample.
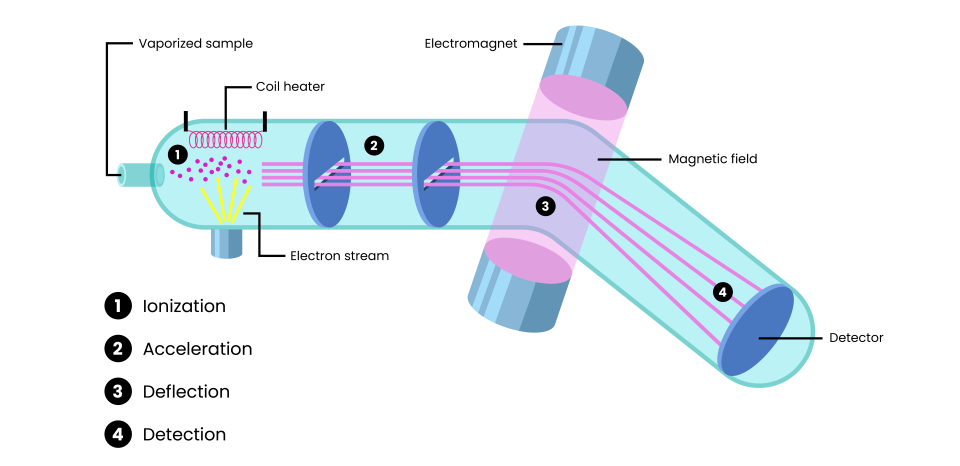
Figure 1. The process of mass spectrometry, including ionization, acceleration, deflection and detection steps.
Types of mass spectrometry
- Quadrupole: What most people think of when they hear ‘mass spectrometry’. It’s the most widely used and most commonly associated with routine applications in clinical diagnostics and pharmaceutical quality control. Quadrupole mass spectrometry uses four parallel rods to create an oscillating electric field that selectively filters ions based on their m/z.
- Most used for: small molecule analysis
- Time-of-flight (TOF): As hinted in the name, TOF MS measures the time it takes for ions to travel through a flight tube to a detector. Lighter ions travel faster than heavier ones, separating them based on their m/z. TOF is known for its high resolution and is often coupled with matrix-assisted laser desorption/ionization (MALDI) for mass spectrometry imaging and tissue analysis.
- Most used for: analyzing large biomolecules
- Electrospray ionization: Generates ions by applying a high voltage to a liquid sample, creating a fine mist of charged droplets. It is commonly used in proteomics, metabolomics and pharmaceutical research due to its ability to ionize large, polar biomolecules (such as proteins, peptides and nucleic acids) without fragmenting them.
- Most used for: high-resolution analysis of large biomolecules
- Ion trap: Captures ions in a 3D electric field inside a cell or along a linear quadrupole, such that scanning ejects ions from the trap according to their m/z.
- Most used for: structural analysis and fragmentation studies of ions

Like having bioanalytical definitions to hand?
Check out the latest Edition of the Bioanalysis Glossary, a free, downloadable resource aiming to keep you up-to-date with the latest terminology and equations within the bioanalytical space.
Mass spectrometry pros and cons
Advantages:
- High sensitivity: detects and quantifies molecules at very low concentrations.
- High specificity: provides precise molecular identification, enabling differentiation between highly similar compounds.
- Versatility: applicable for a wide range of sample types, from small molecules to large, complex biomolecules.
- Analysis of complex mixtures: capable of identifying and quantifying multiple components in a single sample, even in highly complex biological matrices.
- Quantitative & qualitative: offers quantitative concentration data, as well as molecular structure insights.
- Speed: modern machines can analyze samples quickly, making them suitable for high-throughput workflows.
- Coupling with separation techniques: integrates well with chromatography methods.
Disadvantages:
- High cost: expensive to purchase, maintain and operate.
- Complex operation: requires highly skilled personnel, particularly for data interpretation.
- Sample preparation: some techniques demand extensive sample preparation, which can be time-consuming and labor-intensive.
- Limited analysis of non-ionizable compounds: compounds that cannot be ionized effectively will not produce a detectable signal, leading to inaccurate or incomplete analysis. Examples include: non-polar compounds, volatile compounds, or stable/neutral molecules that resist ionization.
- Matrix effects: biological matrices can interfere with ionization.
- Instrument fragility: environmental conditions such as temperature and humidity can affect machine performance, meaning they require regular, careful maintenance.
- Limited field use: most machines are bulky and require controlled lab environments, although smaller, portable machines are emerging.